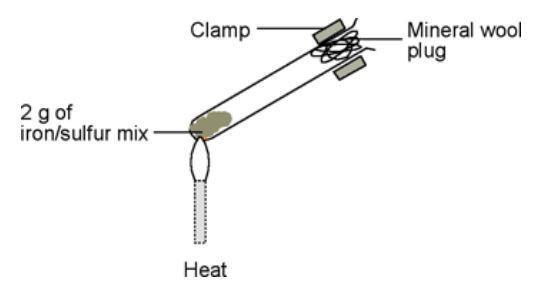
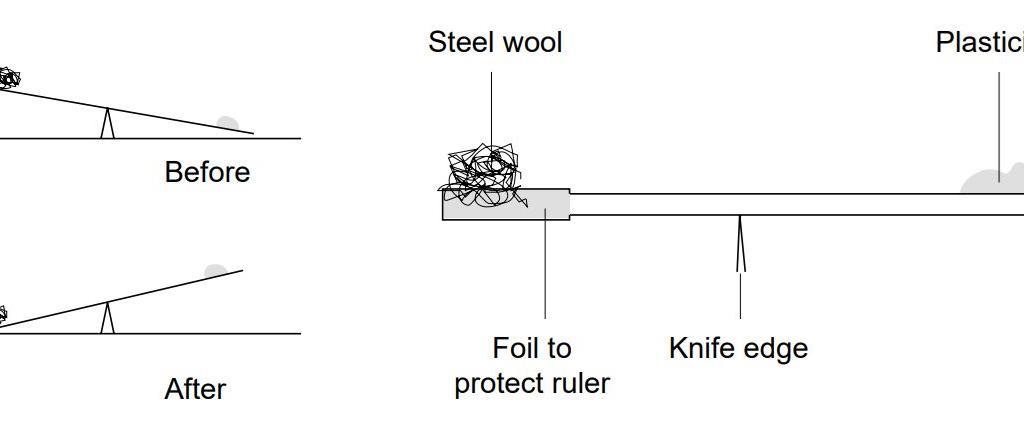
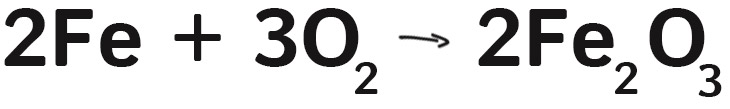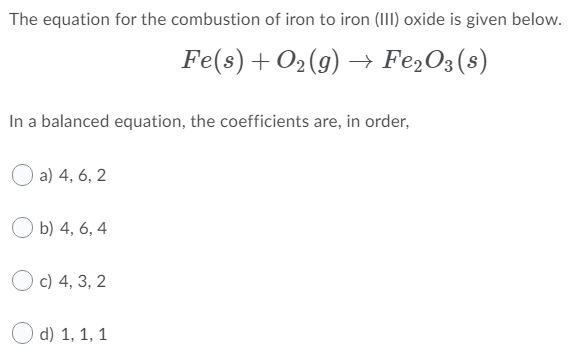SOLVED: In this reaction, iron (steel wool) is combining with oxygen to form iron (III) oxide. Write the balanced equationfor shio reantion; Fe + 0 > 2 Fe3o 4 fe + 501 >

Learning Target: Construct and revise an explanation for the outcome of a simple chemical reaction based on the outermost electron states of atoms, trends. - ppt download

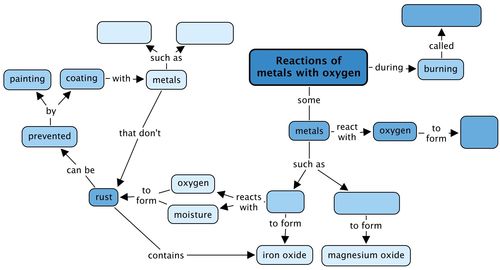
SOLVED: Qbservation: ritc the balanced chemical equation for the combustion of steel wool (iron), the product is iron(II) oxide: B.4 Did bolh bottles test for oxygen? Skip Section B.4 in Summer 2020

Development and characterization of magnetic iron oxide nanoparticles using microwave for the combustion reaction ignition, as possible candidates for biomedical applications - ScienceDirect
ACP - Iron from coal combustion particles dissolves much faster than mineral dust under simulated atmospheric acidic conditions
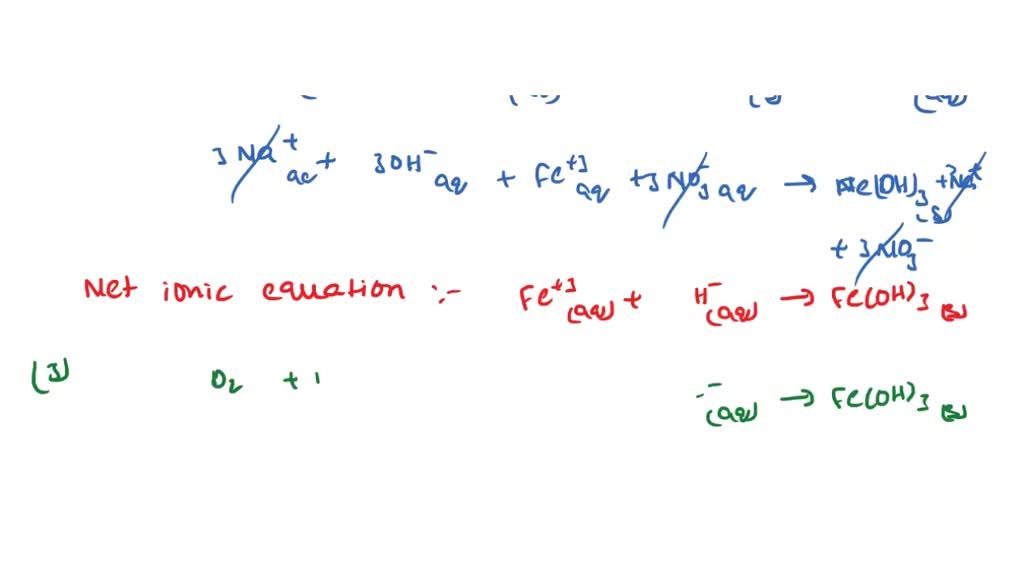
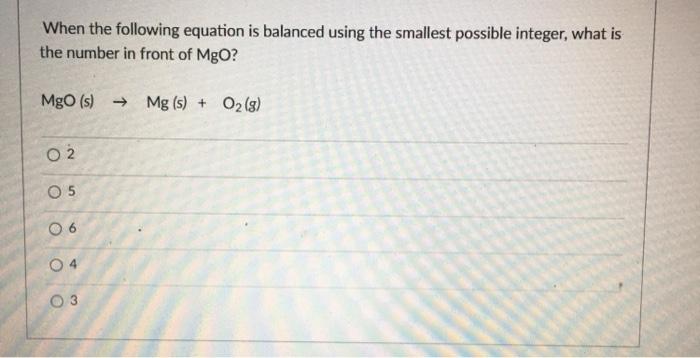


![Incredible steel wool combustion reaction [4K] - YouTube Incredible steel wool combustion reaction [4K] - YouTube](https://i.ytimg.com/vi/Z2llRM6UYlU/maxresdefault.jpg)