
Microbiology Research | Free Full-Text | Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota

Iron Supplements and Probiotics: What You Need to Know, According to a Gut Health Dietitian | Jenna Volpe, RDN, LD, CLT- Functional Dietitian and Clinical Herbalist
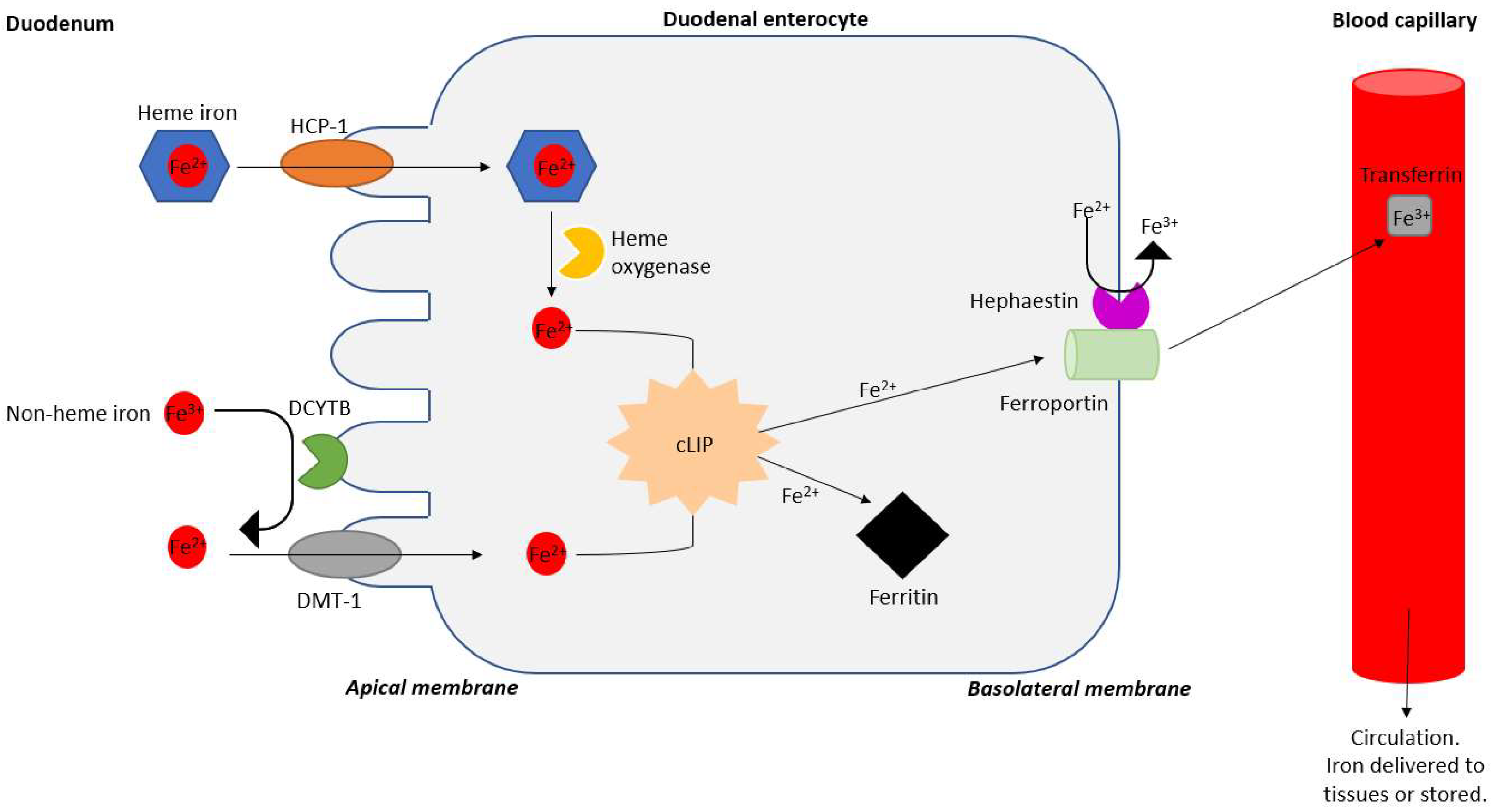
Microbiology Research | Free Full-Text | Oral Iron Supplementation—Gastrointestinal Side Effects and the Impact on the Gut Microbiota

A novel nano-iron supplement versus standard treatment for iron deficiency anaemia in children 6–35 months (IHAT-GUT trial): a double-blind, randomised, placebo-controlled non-inferiority phase II trial in The Gambia - eClinicalMedicine


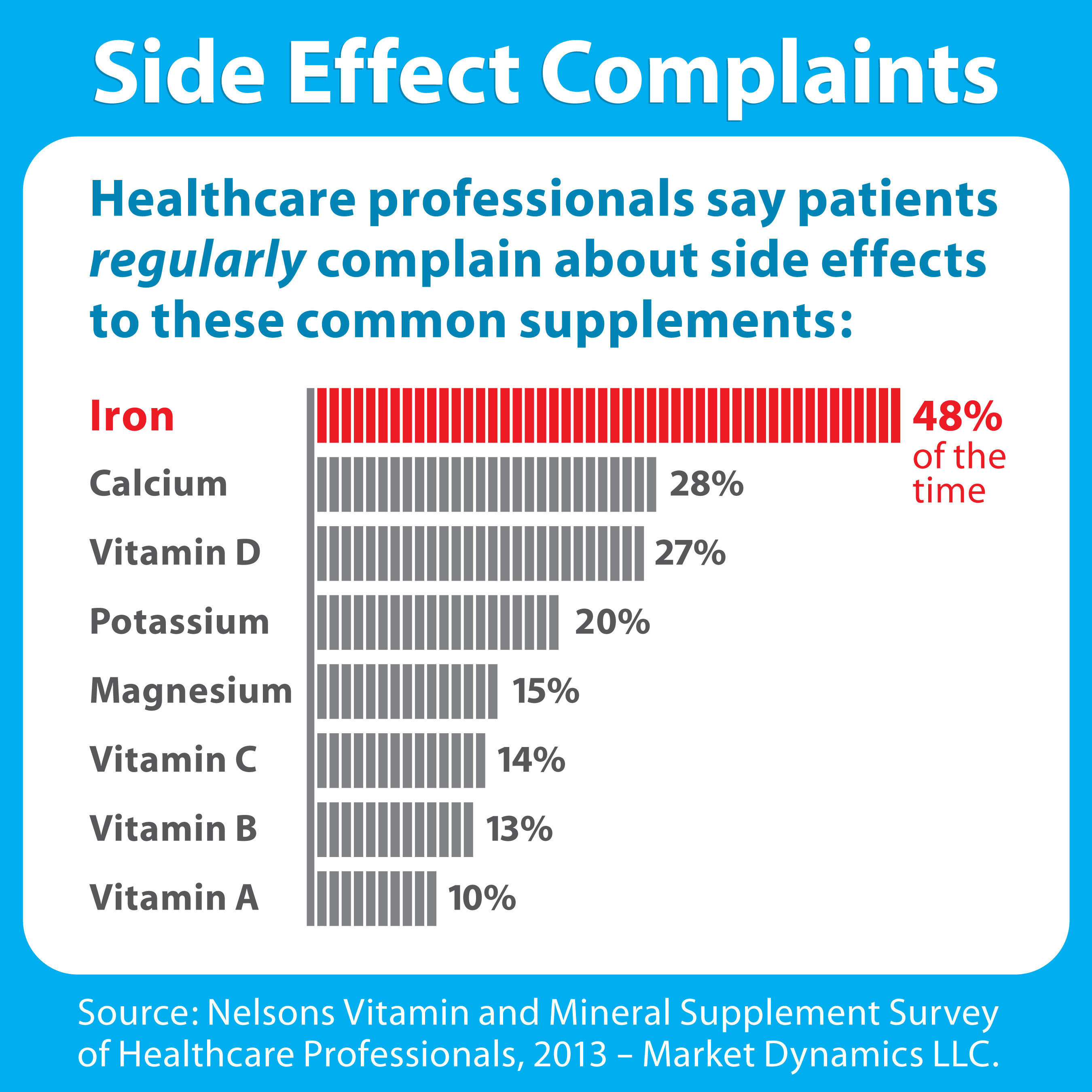
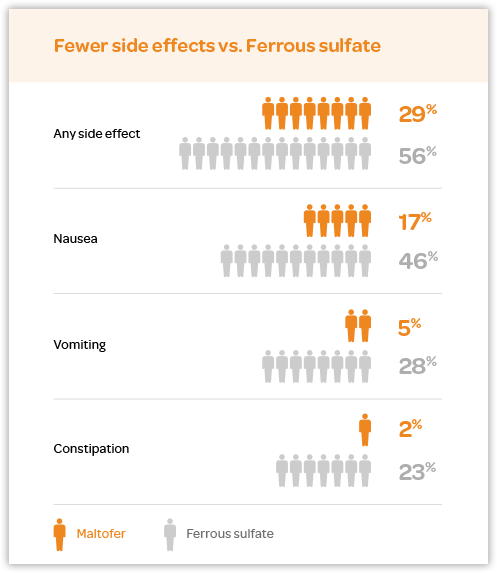










:max_bytes(150000):strip_icc()/do-iron-supplements-for-anemia-cause-upset-stomach-3522504-01-a8806fc982ab45998d75000e183bc406.png)




:max_bytes(150000):strip_icc()/chelated-iron-benefits-4684057_final_HL-c162a71fac594779b62ef8aed927234d.png)
