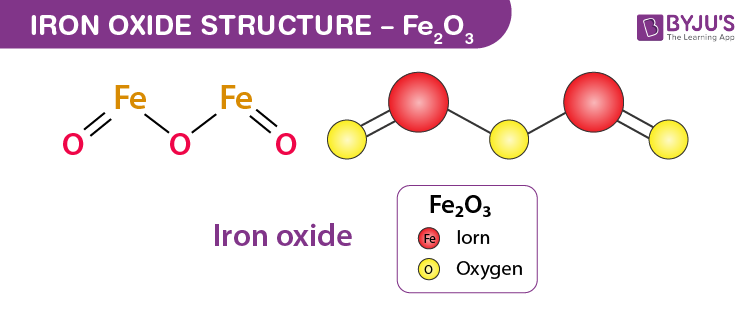
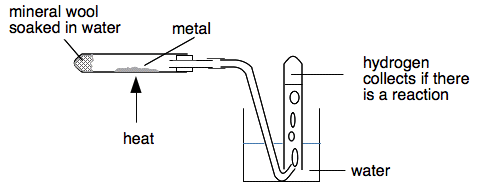
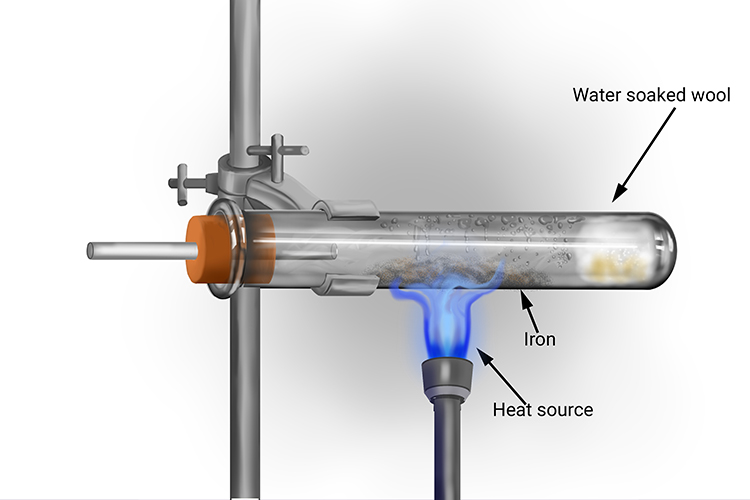
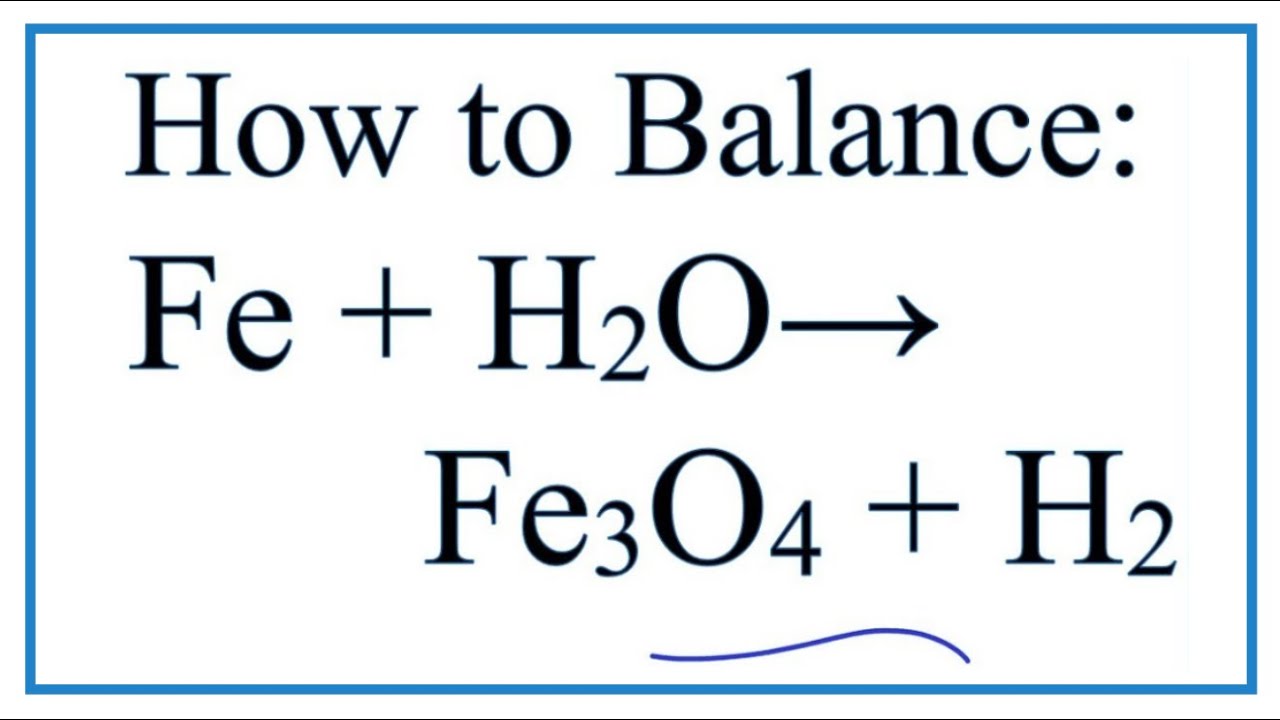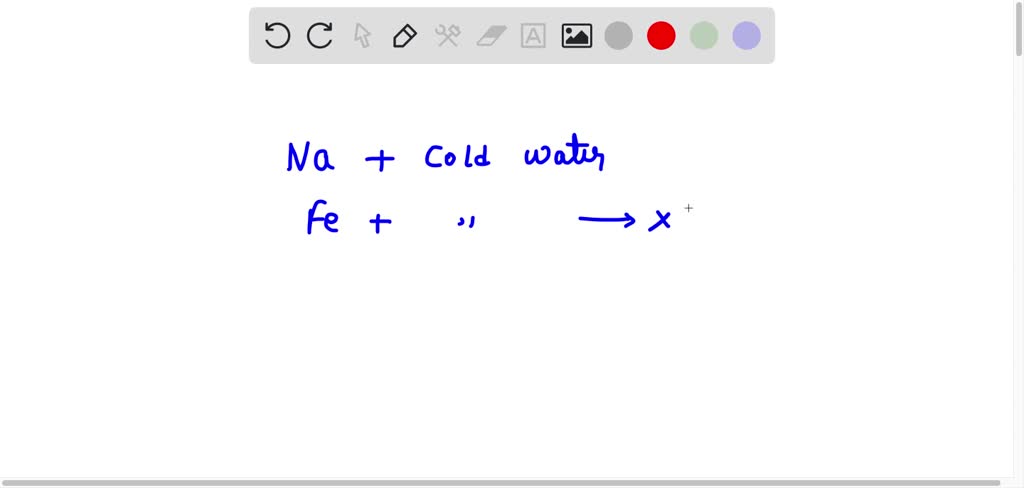Explain the reactions of different metals with hot water, cold water and steam - CBSE Class 10 Science - Learn CBSE Forum

Substituting Chromium in Iron-Based Catalysts for the High-Temperature Water–Gas Shift Reaction | ACS Catalysis

SOLVED:The corrosion (rusting) of iron in oxygen-free water includes the formation of iron(II) hydroxide from iron by the following reaction: Fe(s)+2 H2 O(l) ⟶Fe(OH)2(s)+H2(g) If 1 mol of iron reacts at 298
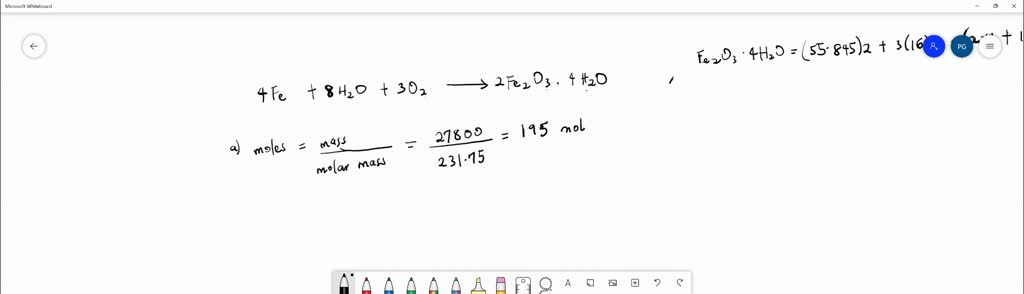
SOLVED: Iron reacts slowly with oxygen and water to form a compound commonly called rust (Fe2O3 · 4H2O). For 27.8 kg of rust, calculate the following quantities. The moles of compound: The

Iron-Based Catalysts for the High-Temperature Water–Gas Shift (HT-WGS) Reaction: A Review | ACS Catalysis

What happen when iron react with water - Science - Metals and Non-metals - 13816401 | Meritnation.com

Iron (III) oxide in water and in hydrochloric acid - Stock Image - C036/3133 - Science Photo Library

i Under what conditions iron reacts with water. ii Give the balanced equation of the reaction. iii What is noticed if the products are not allowed to escape ?

CHARACTERISTICS OF METALS & NON-METALS ACCEPT THE CHALLENGE, SO YOU MAY FEEL THE EXHILARATION OF VICTORY. GEORGE S. PATTON Ch 17. J.C. Rowe Windsor University. - ppt download

Reactivity of Metals | Reactions with Water, Oxygen & Dilute Acids - Video & Lesson Transcript | Study.com

Question Video: Identifying the Balanced Symbolic Equation for the Reaction of Iron Metal with Concentrated Sulfuric Acid | Nagwa

Write equations for the reactions of (i) iron with steam (ii) calcium and potassium with water... - YouTube