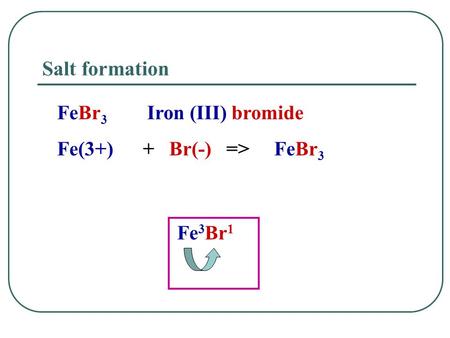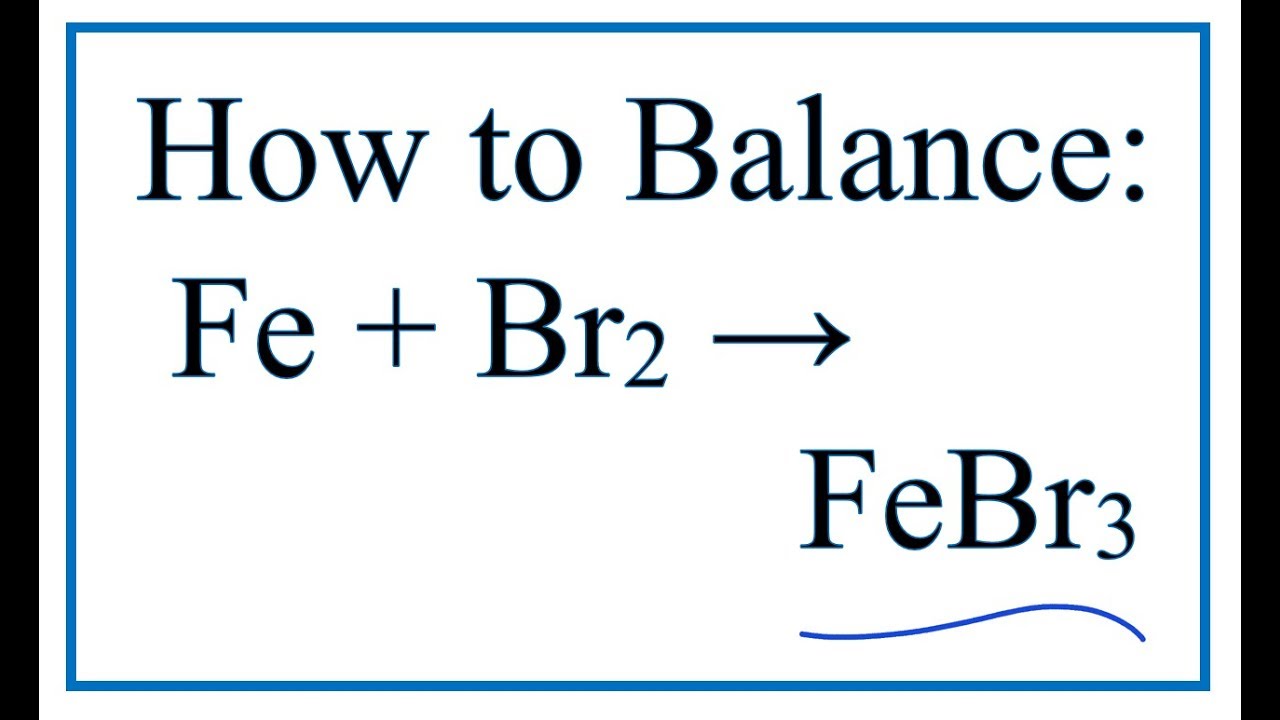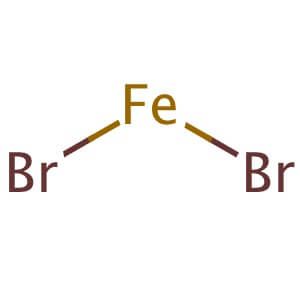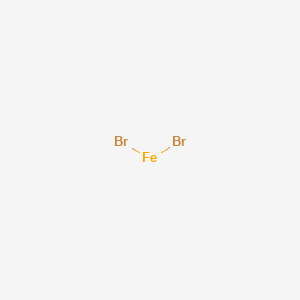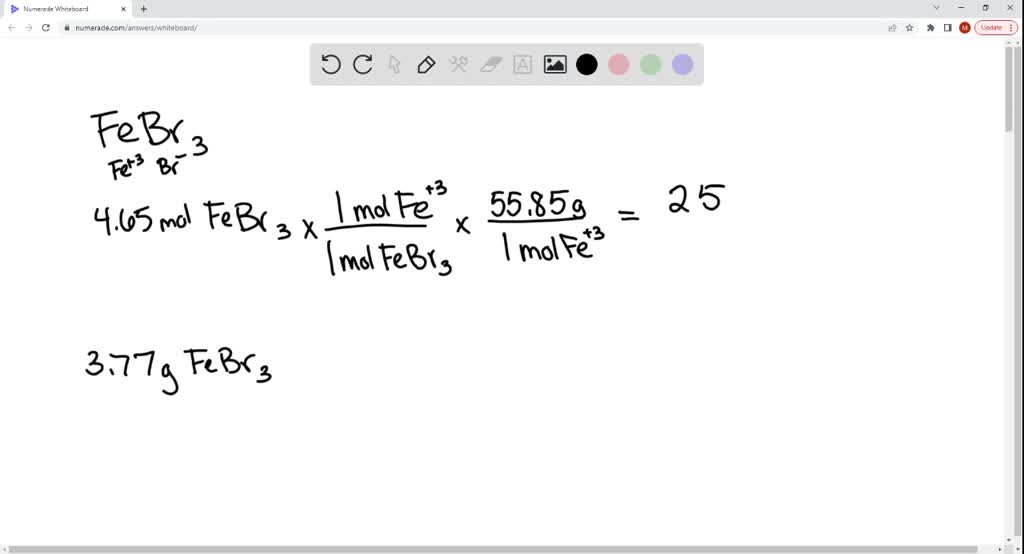
SOLVED: The formula for iron(III) bromide is FeBr3. 1. How many GRAMS of Fe3+ are present in 4.65 moles of iron(III) bromide? 2. How many MOLES of Br- ion are present in

SOLVED: Enter an equation for the precipitation reaction that occurs (if any) when solutions of potassium hydroxide and iron(II) bromide are mixed. Express your answer as a chemical equation. Identify all of