
4 Pack ferric Oxide Powder, Black Iron Oxide Powder | Yellow Iron Oxide Powder | red Iron Oxide Powder | Blue Iron Oxide Powder, Each Color 25 g : Amazon.com.au: Home Improvement

Activator H 2 + O 2 H 2 O 1.What is this chemical equation showing the formation of? 2.Do you think this equation is balanced? Why? - ppt download
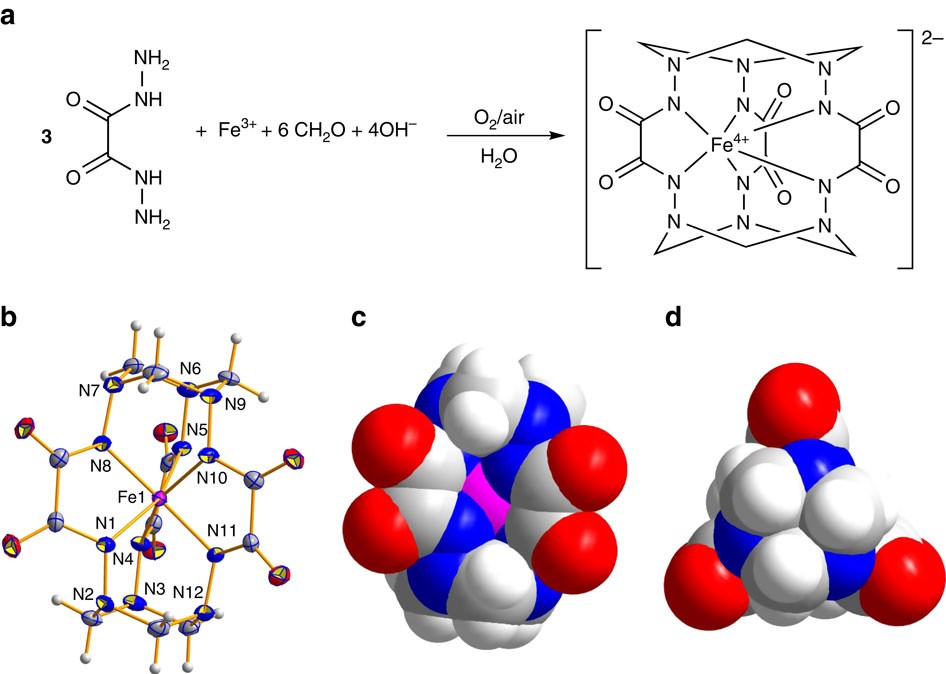
Indefinitely stable iron(IV) cage complexes formed in water by air oxidation | Nature Communications

Iron‐Based Layered Cathodes for Sodium‐Ion Batteries - Gao - 2021 - Batteries & Supercaps - Wiley Online Library
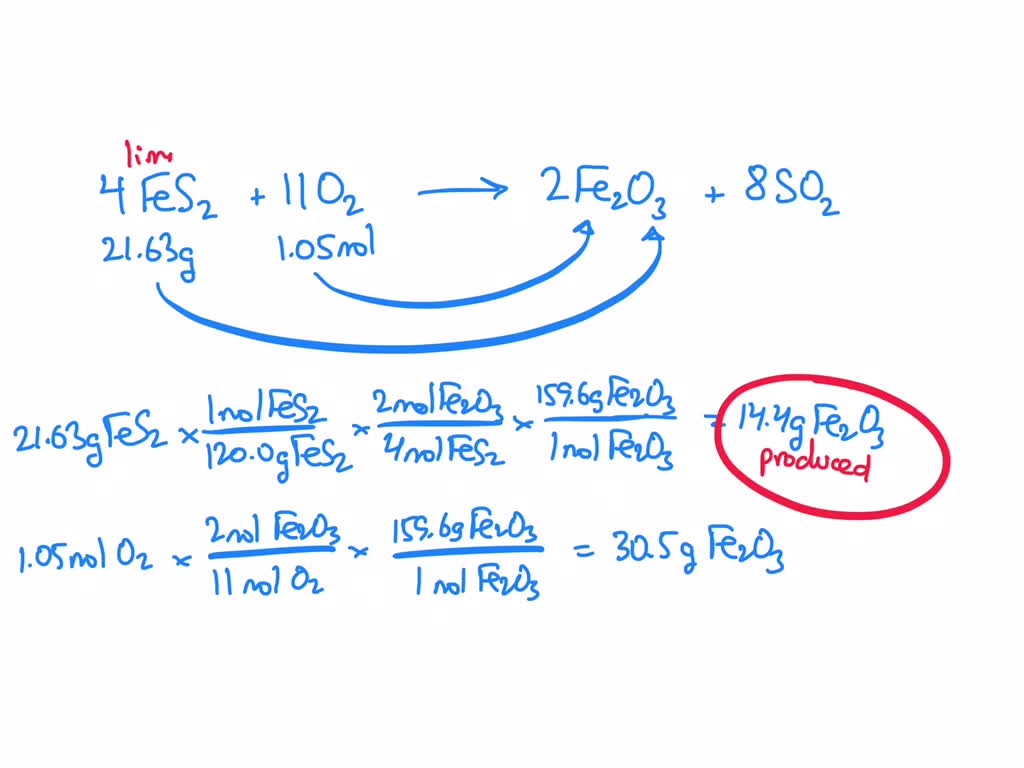
SOLVED: When fool's gold, iron(II) sulfide reacts with oxygen, it forms ferric oxide and sulfur dioxide. If a 21.63-gram nugget of fool's gold is reacted with 1.05 moles of oxygen, what mass

Determine the molecular formula of an oxide of iron in which the mass per cent of iron and oxygen are 69.9 and 30.1 respectively.







![ANSWERED] For the following statements (1) Cast ir... - Inorganic Chemistry ANSWERED] For the following statements (1) Cast ir... - Inorganic Chemistry](https://media.kunduz.com/media/sug-question/raw/68218076-1658826933.790158.jpeg)




