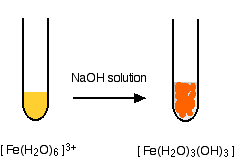
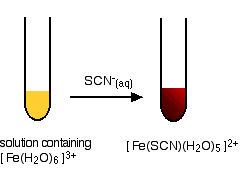
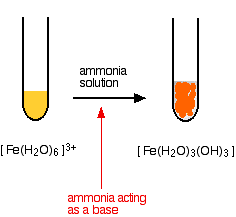
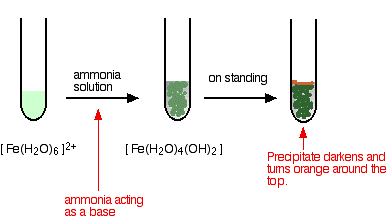
Removal of iron, manganese and ammonia from groundwater using a PAC-MBR system: The anti-pollution ability, microbial population and membrane fouling - ScienceDirect

Catalysts | Free Full-Text | Thermodynamics of Chemical Processes in the System of Nanocrystalline Iron–Ammonia–Hydrogen at 350 °C

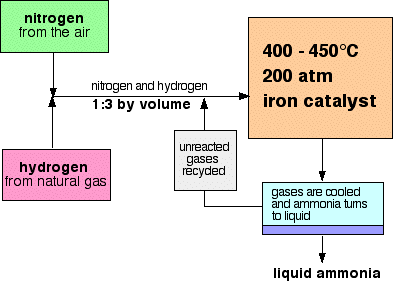
Ammonia Synthesis Production & Reaction | How Is Ammonia Made? - Video & Lesson Transcript | Study.com

Ammonia production using iron nitride and water as hydrogen source under mild temperature and pressure - ScienceDirect

Development and Recent Progress on Ammonia Synthesis Catalysts for Haber–Bosch Process - Humphreys - 2021 - Advanced Energy and Sustainability Research - Wiley Online Library
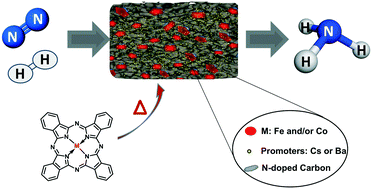
Development of catalysts for ammonia synthesis based on metal phthalocyanine materials - Catalysis Science & Technology (RSC Publishing)
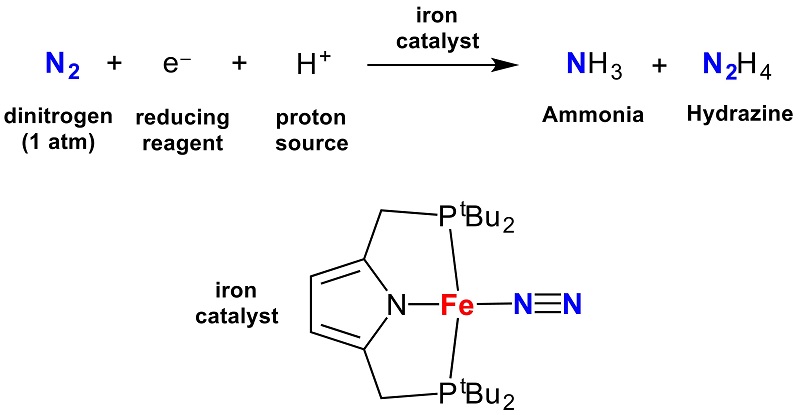
Catalytic reduction of nitrogen gas by cheap iron catalyst under mild conditions | The University of Tokyo
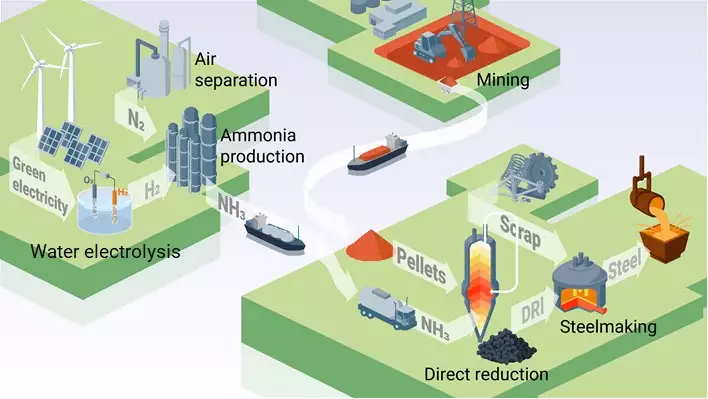
Ammonia, Efficient Hydrogen Carrier and Green Steel Enabler - Max Planck Materials Scientists Use Ammonia for Sustainable Iron- and Steelmaking - Hydrogen Central

Design of Iron-Nickel Nanocatalysts for Low-Temperature Electrochemical Ammonia Generation – Ammonia Energy Association

Scheme of ammonia synthesis on iron catalysts. (a) Initial reaction... | Download Scientific Diagram

Study of the Iron Catalyst for Ammonia Synthesis by Chemical Potential Programmed Reaction Method | The Journal of Physical Chemistry C

Si-Doped Fe Catalyst for Ammonia Synthesis at Dramatically Decreased Pressures and Temperatures | Journal of the American Chemical Society

Chemical Transformation of Fe, Air & Water to Ammonia: Variation of Reaction Rate with Temperature, Pressure, Alkalinity and Iron | Inorganic Chemistry | ChemRxiv | Cambridge Open Engage
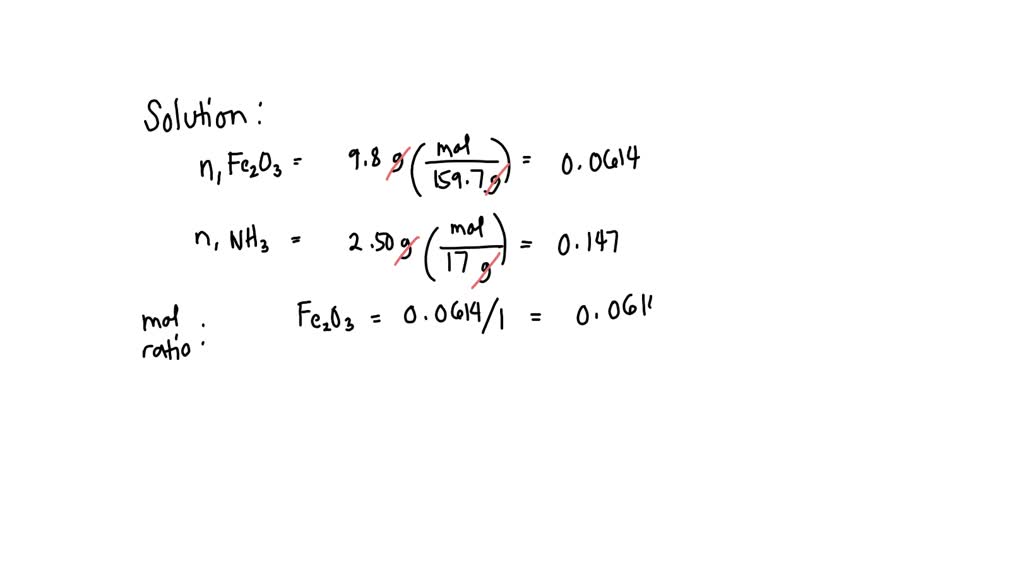
SOLVED: Iron(III) oxide is reacted with ammonia to produce solid iron, gaseous nitrogen, and water. 1 Fe2O3 (s) + 2 NH3 (g) –> 2 Fe (s) + 1 N2 (g) + 3