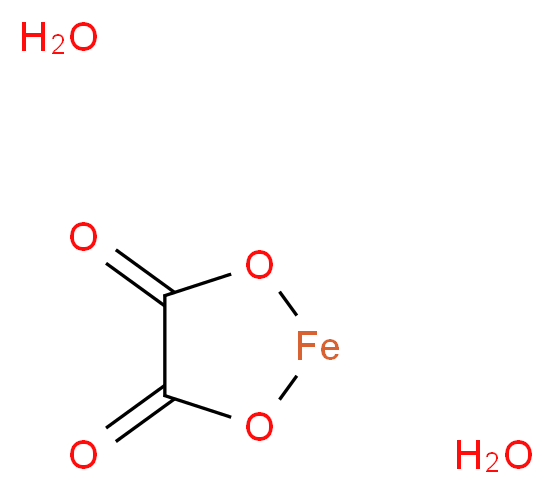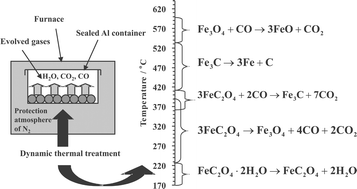
Thermal behaviour of iron(ii) oxalate dihydrate in the atmosphere of its conversion gases - Journal of Materials Chemistry (RSC Publishing)

Minerals | Free Full-Text | Iron(II)oxalate Dihydrate—Humboldtine: Synthesis, Spectroscopic and Structural Properties of a Versatile Precursor for High Pressure Research

Real-Time Studies of Iron Oxalate-Mediated Oxidation of Glycolaldehyde as a Model for Photochemical Aging of Aqueous Tropospheric Aerosols | Environmental Science & Technology

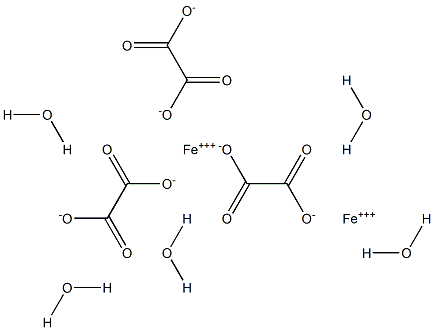
Iron ( II ) oxalate is an interesting inorganic compound with a beautiful yellow color. It is conveniently prepared in the laboratory by the reaction of Fe2+ salts with oxalic acid. It

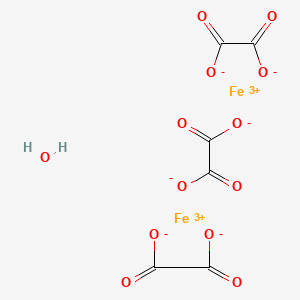
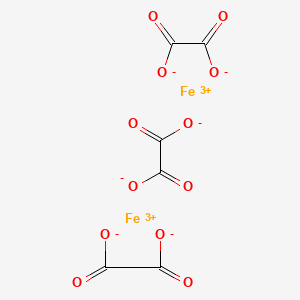
Fate of iron oxalates in aqueous solution: The role of temperature, iron species and dissolved oxygen - ScienceDirect
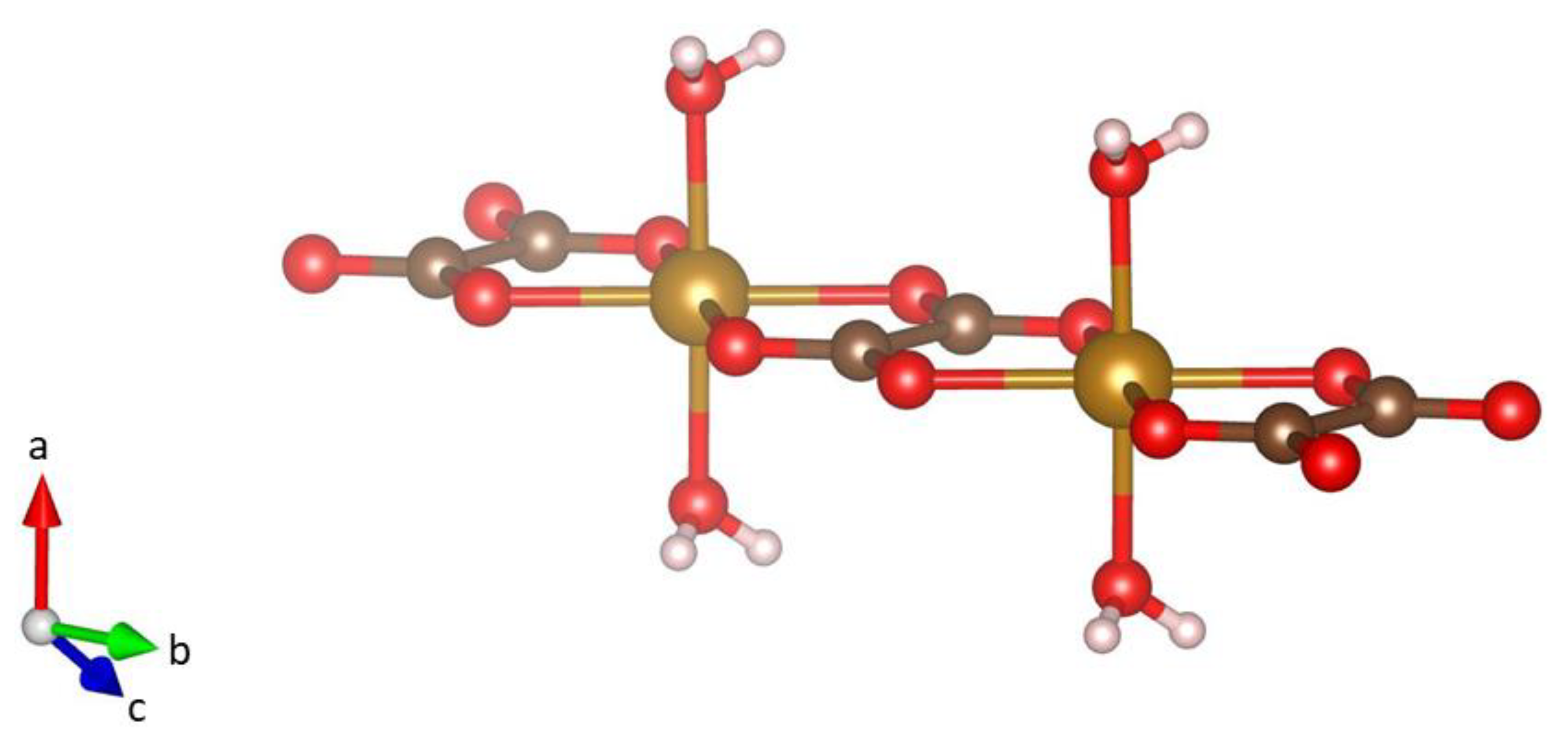
Minerals | Free Full-Text | Iron(II)oxalate Dihydrate—Humboldtine: Synthesis, Spectroscopic and Structural Properties of a Versatile Precursor for High Pressure Research

Real-Time Studies of Iron Oxalate-Mediated Oxidation of Glycolaldehyde as a Model for Photochemical Aging of Aqueous Tropospheric Aerosols | Environmental Science & Technology